 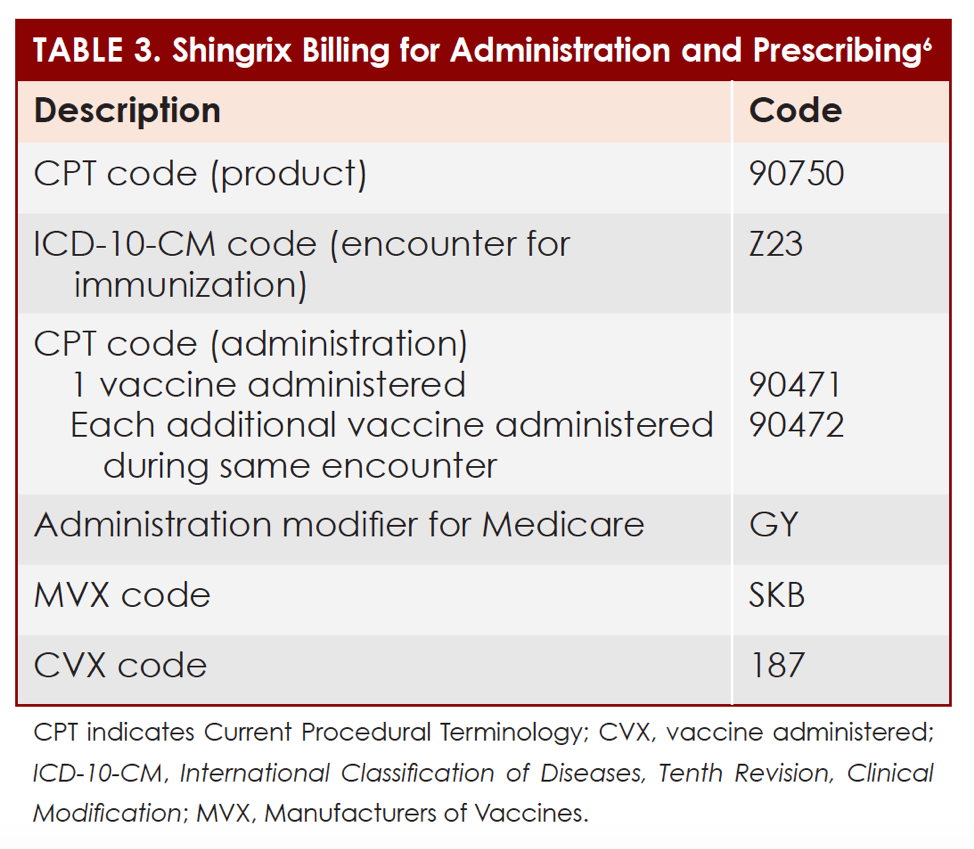
Postherpetic neuralgia is a common type of nerve pain that arises with shingles. In addition, they are not used to prevent primary varicella infection, also known as chickenpox. Shingrix and Zostavax are approved for preventative purposes only. However, unlike Zostavax, Shingrix is also approved to prevent shingles in immunocompromised or immunodeficient adults aged 18 years and older. Both vaccines are indicated to prevent shingles in adults aged 50 years and older. Shingrix and Zostavax are FDA-approved to prevent shingles (herpes zoster). Vaccination is complete in 2 to 6 months after 2 dosesĬonditions treated by Shingrix and Zostavax Main differences between Shingrix and ZostavaxĪdminister 1 dose (0.5 ml) into the muscle and then another dose (0.5 ml) between 2 and 6 months laterĪdminister one dose (0.65 ml) once under the skin Zostavax is no longer available in the U.S. The frozen version must be kept frozen during transport and storage to ensure its effectiveness, while the refrigerator-stable Zostavax can be kept in a refrigerator until it needs to be used. It comes in a frozen version and a refrigerator-stable version. Zostavax is administered as a single injection underneath the skin (subcutaneous). Otherwise, the vaccine may cause a symptomatic infection, even weeks following vaccination. For this reason, Zostavax is not recommended for those who are immunocompromised. In other words, Zostavax contains a weakened version of the actual virus to produce an immune response. 
Zostavax, approved in 2006, is a live, attenuated herpes zoster vaccine. Shingrix is currently the preferred shingles vaccine, according to the Centers for Disease Control and Prevention (CDC). Immunocompromised or immunodeficient individuals may require a shorter vaccine schedule and can get the booster one to two months after the first shot. The second dose is necessary to ensure long-term effectiveness. It is given in two separate doses with a period of two to six months between the first and second doses. Shingrix is administered as an injection into the muscle (intramuscular). Because Shingrix is an inactivated or non-live vaccine, it can be used in immunocompromised patients or those with a weakened immune system. The adjuvant suspension in Shingrix contains an extract from the Quillaja saponaria tree, known to modulate immune activity. Shingrix also contains an adjuvant, or added ingredient, to help boost the body’s immune response to the virus. It contains a certain protein called the varicella-zoster glycoprotein E (gE) antigen to produce an immune response in the body. Shingrix (What is Shingrix? ) is a recombinant, adjuvanted zoster vaccine that was first FDA-approved in 2017. What are the main differences between Shingrix and Zostavax? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
